Leverage uBriGene’s expertise in viral vector analytical development, assay validation, and final product release to ensure superior quality and compliance.

As a leading viral vector CDMO, uBriGene has successfully manufactured and released over 60 batches of viral vectors, including AAV, LVV, adenovirus, and oncolytic viruses. Partner with our experienced viral vector analytics team to ensure the quality and safety of your cell and gene therapy products.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ensure the safety, quality, and regulatory compliance of your viral vector products with uBriGene’s expert analytical development and QC testing services.
Contact the ExpertsExtensive experience
60+ GMP viral vector batches successfully delivered
Strong regulatory support

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
The uBriGene team has validated a comprehensive suite of assays for LVV analytics, supporting both process development and final product lot release. Additionally, our analytics development team collaborates with clients to develop and validate custom product-specific assays. We also routinely perform tech transfers for assay methods not offered in-house but preferred by our clients.
Analytical and QC testing categories for lentivirus:
The advantages of membrane matrix in downstream chromatography for plasmid purification include requiring only 30% of the time compared to conventional resin. Therefore, plasmid DNA is manufactured faster and at a much lower cost.
The uBriGene team has a rich history supporting AAV clinical trials for programs focused on diseases of the eye, CNS, and blood. As a result of that extensive experience, we have developed an impressive collection of orthogonal assays for addressing the multiple challenges of consistently producing and characterizing high quality and high titer AAV drug products. This includes robust assays for the critical, but often challenging, quantitative assessment of empty/full capsid ratios.
Analytical and QC testing categories for AAV:
Quality control (QC) testing is a critical component in the production of adenoviral vectors to ensure they meet rigorous safety, efficacy, and regulatory standards. Comprehensive QC testing evaluates key attributes such as identity, potency, purity, and stability, ensuring the adenovirus product is suitable for clinical and commercial use. Below is a list of essential assays used in adenovirus QC testing to guarantee consistent product quality and therapeutic success.
Analytical and QC testing categories for adenovirus:
Safety: Endotoxin, sterility, mycoplasma
The uBriGene team has a track record of success manufacturing a variety of oncolytic viral vectors, including vaccinia virus. The testing of these vectors requires rigorous quantitative assessment of the potency and specificity of tumor cell killing. The uBriGene team has the established expertise to efficiently design, validate, and execute those important functional tests.
Analytical and QC testing categories for oncolytic viruses:
AAV is known to assemble empty capsids during production, which can impact efficacy and increase immunogenicity. Accurate full/empty ratio testing is critical for AAV drug products.
The current gold standard for empty/full ratio testing is analytical ultracentrifugation (AUC), which provides comprehensive information on the components of AAV products, including the percentages of aggregates, full, partial, and empty capsids.

Figure 1. AUC analysis of AAV full/empty capsid ratio.
The infectious titer or transduction titer is often much lower than the genomic titer. Therefore, infectious/transduction titer testing is a critical QC assay for the release of GMP-grade lentiviral vector batches.
Two main methods are commonly used, depending on the application and required sensitivity. The first is an integration and qPCR-based method that measures genomic integration after transducing cells with the lentiviral vector. The second is a functional titration, protein expression-based method. For CAR-T applications, flow cytometry is typically employed, using antibody staining to detect the expressed CAR after lentiviral vector transduction.
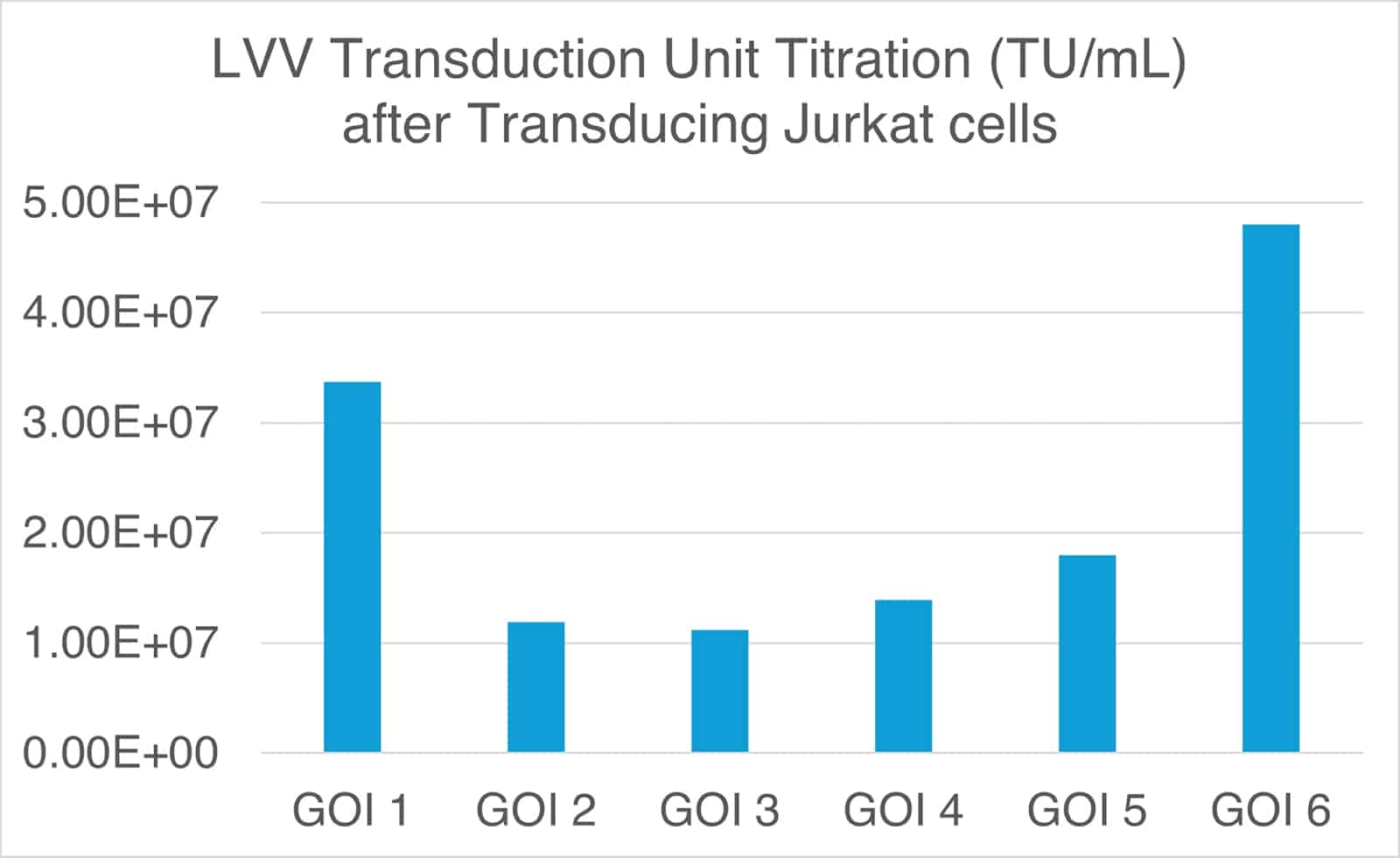
Figure 2. Transduction titration of lentiviral vectors after transducing Jurkat cells. Lentiviral vectors encoding six different genes were manufactured using uBriGene’s suspension cell line, 293TH. Flow cytometry was used to detect LVV-transduced cells, and the transduction titer was calculated.
Unveil the current manufacturing approaches for LVV and its deployment in cell therapies.
Discover our AAV manufacturing capabilities, scalable processes, and testing platform.
Explore uBriGene's extensive expertise in ATMP manufacturing.
Several methods are commonly used to assess the empty-to-full capsid ratio of AAV vectors, including optical density measurement (OD260/280), transmission electron microscopy (TEM), analytical ultracentrifugation (AUC), and HPLC.
Among these, AUC is considered the "gold standard" for measuring AAV empty/full capsid ratios. AUC separates particles based on their sedimentation coefficients, offering high-resolution, sensitive, and quantitative analysis. It provides detailed profiles of capsid populations, distinguishing empty, partial, and full capsids, without requiring labels or stains.
At uBriGene, multiple methods are available for analyzing AAV empty/full ratios; however, AUC is the preferred method for release testing due to its superior accuracy and reliability. The full particle ratio of AAV products manufactured by uBriGene typically ranges from 80% to 95%, depending on the serotype.
Yes, uBriGene performs the AAV TCID50 assay to determine the infectious titer of Adeno-Associated Virus (AAV). This process involves culturing HeLa RC32 cells in a 96-well plate, followed by the addition of serial dilutions of the AAV sample to allow for infection.
After incubation, infection is detected using primers targeting the gene of interest sequence and conducting qPCR to assess positive or negative results. The TCID50 value is then calculated using the Reed-Muench or Spearman-Karber method, based on the dilution at which 50% of the wells are infected.
The replication-competent AAV (rcAAV) assay is essential to ensure the safety of AAV vectors by detecting the presence of rcAAV. This is typically performed using cell-based assays with HEK293 or HeLa cells. The cells are infected with the AAV sample, along with a helper virus (e.g., adenovirus) to facilitate AAV replication.
Detection of rcAAV is then carried out using qPCR or by observing cytopathic effects. uBriGene offers rcAAV testing, with a release standard of less than 1 rcAAV per 1E8 AAV particle.
Detecting adventitious viruses in vaccinia virus products is challenging due to the virus's ability to infect and lyse most cells and the absence of highly effective neutralizing antibodies. Currently, Next-Generation Sequencing (NGS) is the primary method used for detection.
Next-Generation Sequencing (NGS) offers unparalleled advantages for detecting adventitious viruses, including comprehensive and unbiased screening of all nucleic acids in a sample. Its high sensitivity and specificity allow for the detection of low-level contamination with precise sequence identification, minimizing false positives and negatives.
Ensuring that HCD residual fragments in GMP-grade lentiviral vector production are under 200 base pairs (bp) is critical for several reasons. It minimizes the risk of oncogenesis by reducing the likelihood of integration into the host genome, ensures compliance with regulatory guidelines on residual DNA size and quantity, and mitigates immune responses while maintaining high purity standards.
To monitor HCD fragment sizes, uBriGene utilizes capillary electrophoresis (CE), ensuring no fragments exceed 200 bp. These measures collectively enhance the safety, purity, and efficacy of lentiviral products for clinical use.
As an enveloped virus, lentiviral vectors are prone to losing infectivity. Formulation studies are essential to extend their shelf life. For clinical applications, storage and stability studies are critical to ensuring the safety and efficacy of lentiviral vector drug products or other critical materials.
Partner with our viral vector experts to ensure the safety, efficacy, and regulatory compliance of your therapeutic programs.
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Request a quote
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ask the plasmid DNA expert! With over 300 GMP batches in our track record and our cost-effective platform technology, we can help accelerate your clinical programs.