
uBriGene’s LVV Turbo™ lentivirus production platform integrates three cutting-edge technologies to deliver higher-yield, high-potency lentivirus at lower cost.
Download Brochure
FDA-registered cGMP cell therapies, fully compliant, traceable, ready-to-use, and backed by 10+ years of proven manufacturing excellence.

Learn how cutting-edge technologies to alleviate lentiviral supply constraints, reduce manufacturing costs, and support seamless scale-up from early development through commercial readiness.

Delivering high-quality TIL manufacturing globally, uBriGene Biosciences combines automation, scalability, and precision with over 10 successful GMP batches.

Learn how uBriGene can deliver high efficiency total solution designed to accelerate the development of iPSC-derived cell therapies.
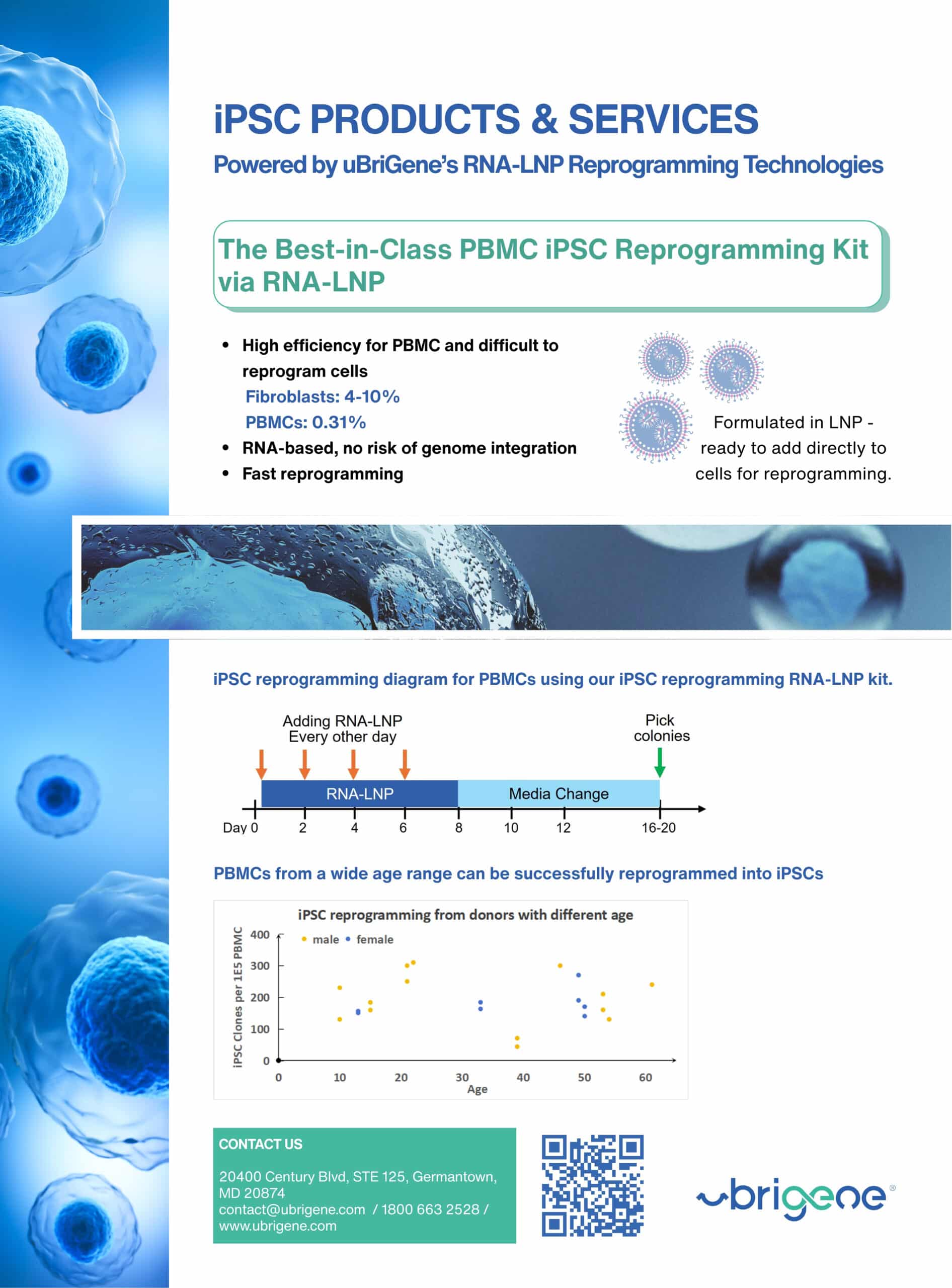
Unlock safer regenerative therapies with uBriGene’s Genome-Safe iPSC reprogramming built for clinical success.

Redefining RNA Therapeutics: GMP RNA-LNP Manufacturing You Can Trust

Explore how uBriGene's cutting-edge solutions can fast-track iPSC-based therapeutic development, enabling the next generation of disease models and cell therapies.

Explore the fundamentals of RNA drug development and the critical role of in vitro transcription (IVT) in manufacturing workflows.

Learn how uBriGene can be your bridge from ATMPs concept to commercialization.

Explore uBriGene’s robust GMP plasmid manufacturing process and testing capabilities.