GMP LVV Turbo™ lentivirus manufacturing platform with access to qualified raw materials that meet the highest regulatory standards for lentiviral vector production.
Our GMP lentivirus production platform, LVV Turbo™, is a mature, third-generation system designed to accelerate lentiviral vector development.
It features our high-productivity 293TH suspension cell line, a fully closed downstream purification process, and the Ultra-T enhancer technology through our partnership with Syenex. The platform also provides clients with access to lentiviral helper plasmids, which are accompanied by Drug Master Files (DMFs) on file with the FDA, enabling faster and more efficient GMP lentivirus manufacturing.
Leverage our high-productivity LVV Turbo™ lentivirus production platform for effective and cost-saving GMP lentivirus manufacturing.
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Contact the Experts
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
GMP grade
Activated DMF on file with the FDA
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Contact the Experts
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
On-demand Webinar:
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Watch NowuBriGene’s LVV Turbo™ lentivirus production platform integrates three cutting-edge technologies – our high-productivity 293TH suspension cell line, a fully closed downstream purification process, and the partnered Ultra-T enhancer – to deliver higher yield, higher potency, and lower cost.
Together, as a lentivirus CDMO, these innovations enable the LVV Turbo™ GMP lentivirus production platform to significantly reduce the cost of goods for CAR-T therapies while improving performance.
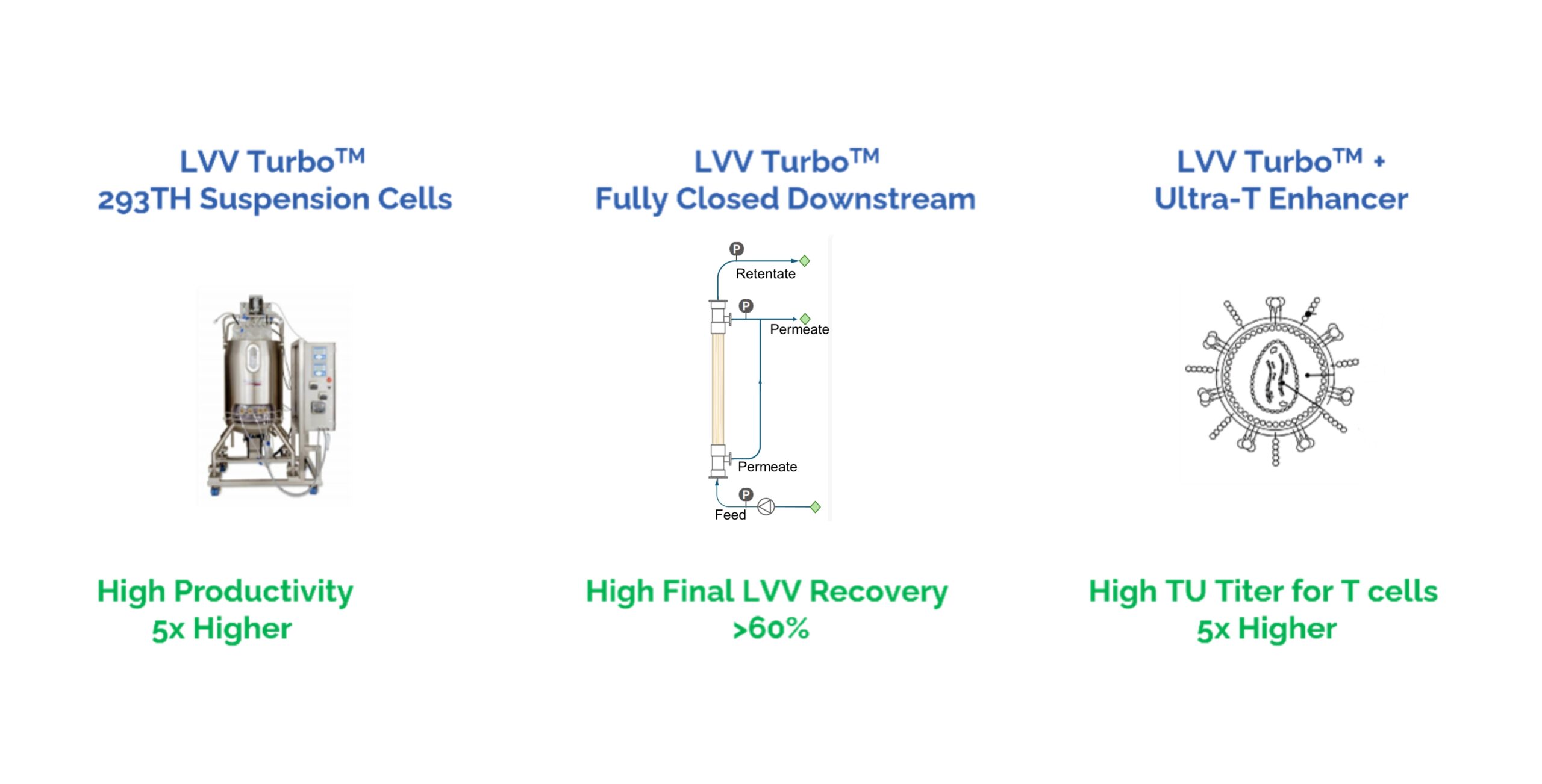
Fig. 1. Diagram of the three disruptive technologies that power our LVV TurboTM platform.
 Request a quote
Request a quoteuBriGene offers a full suite of in-house and custom lentivirus-specific assays to support lentiviral vector process development and manufacturing, including QC release and stability indicating test methods.
| Test Item | Method | |
|---|---|---|
| Identity | Sanger sequencing* | |
| Physical property | Appearance | Visual inspection |
| Chemical property | pH | pH measurement |
| Content | Physical titer by p24 | ELISA |
| Infectivity | LTI calculation | |
| Transduction titer | Flow cytometry | |
| Purity | Residual host DNA | qPCR |
| Residual plasmid DNA | qPCR | |
| Residual host protein | ELISA | |
| Residual Benzonase | ELISA | |
| Residual host DNA (SV40 Large T antigen) | ddPCR | |
| Residual host DNA (E1A) | ddPCR | |
| Safety | Endotoxin | Gel-clot assay |
| Sterility | Membrane filtration method | |
| Mycoplasma | Culture method | |
| RCL (EOPC & UPB) * | Culture method | |
*Outsourced testing
Rhoncus duis elementum suspendisse odio amet arcu tellus. Sapien felis vestibulum euismod eget orci scelerisque id. Non ut sagittis leo et. Et pretium nisl ultrices dapibus sit euismod volutpat elementum.
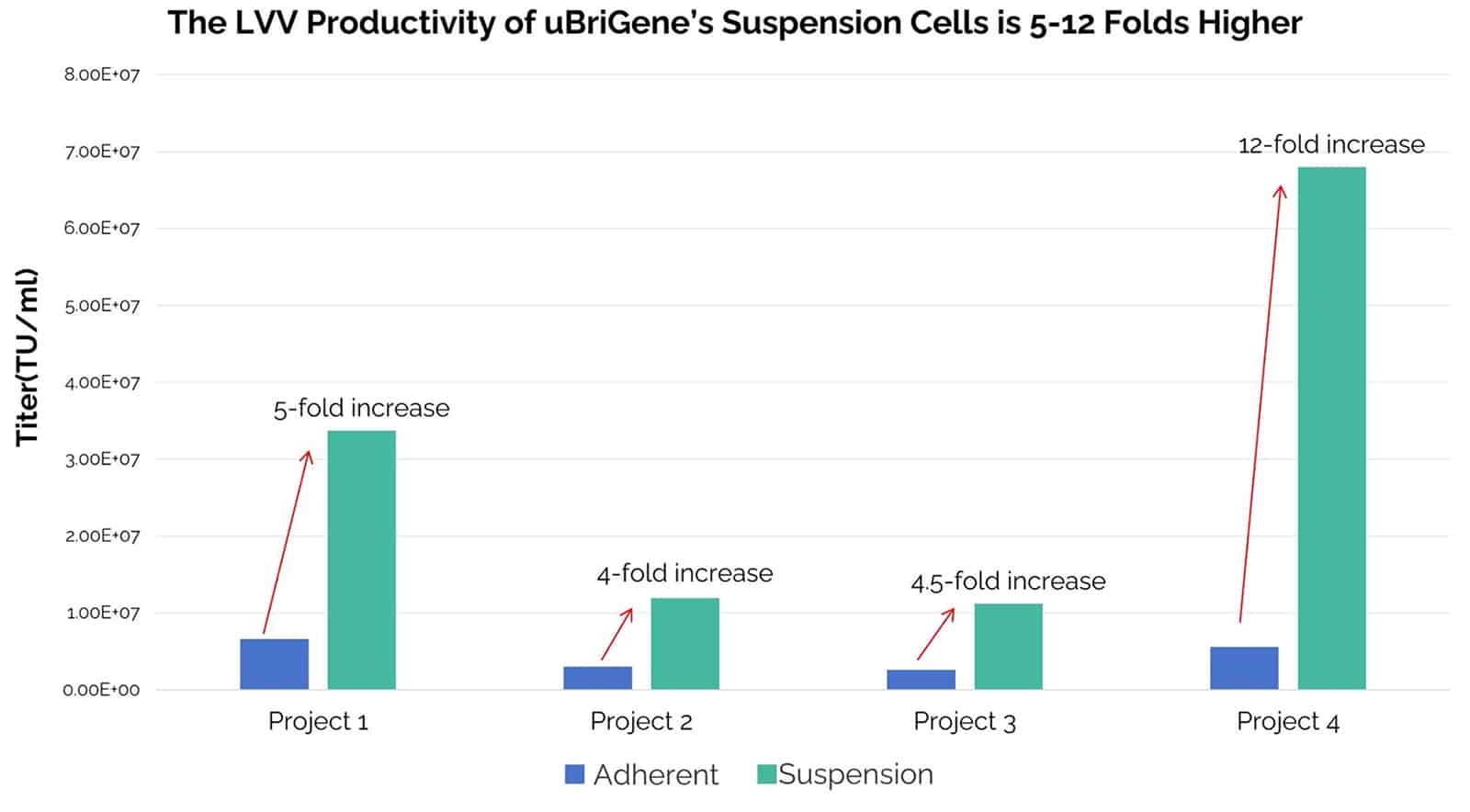
Fig. 2. Comparison of lentivirus productivity of HEK293T adherent cells and uBriGene’s 293TH suspension cells. Four different GOIs were used for this study. The transduction units were analyzed using flow cytometry after transducing Jurkat cells with the lentiviral vectors produced from HEK293T adherent cells and our suspension 293TH cells.
Request a quote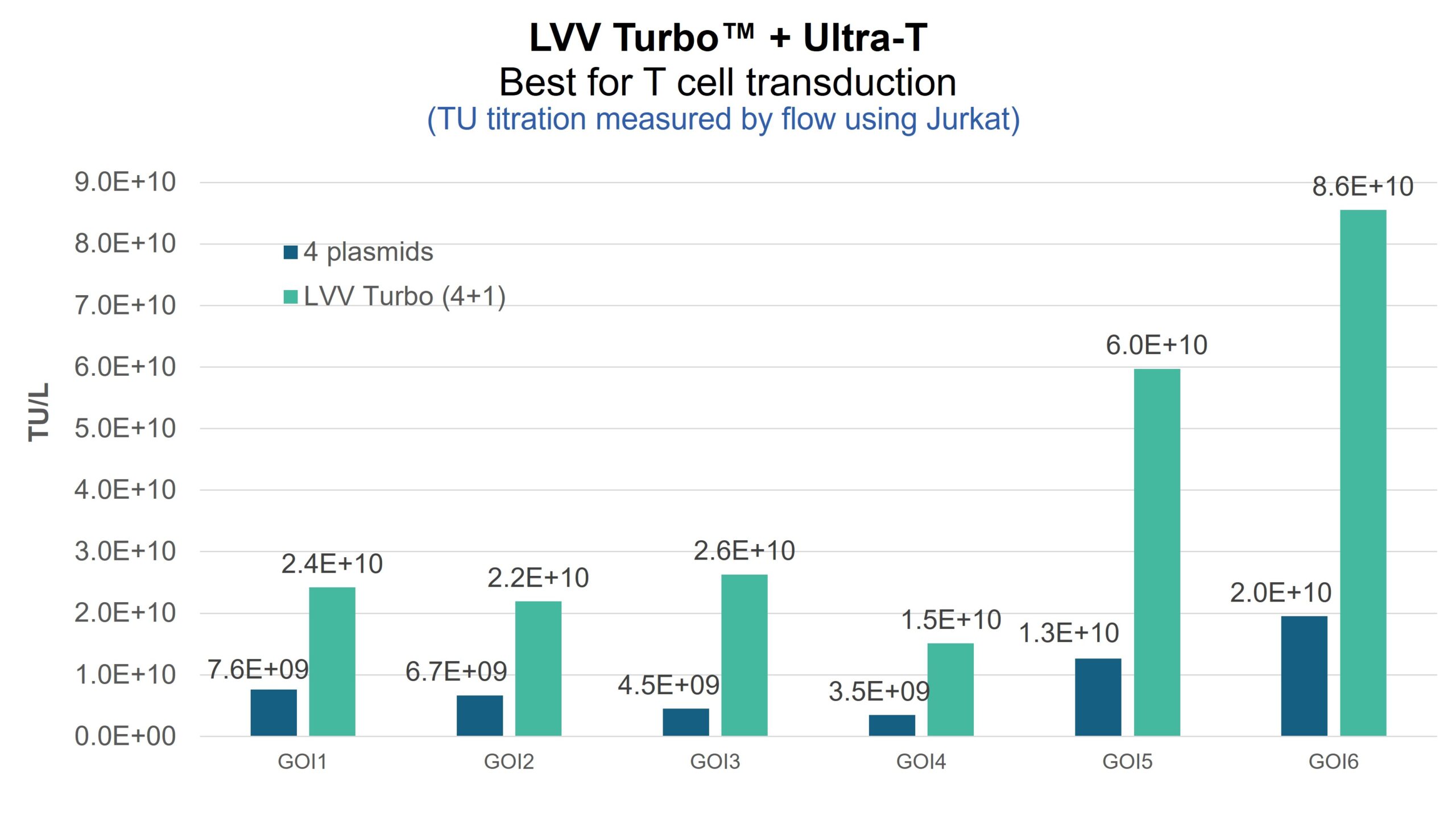 Request a quote
Request a quoteFig. 3. The figure compares LVV transduction titers produced with the standard 4-plasmid system versus the LVV Turbo™ + Ultra-T System, showing up to a 5× increase in functional titer across six GOIs.
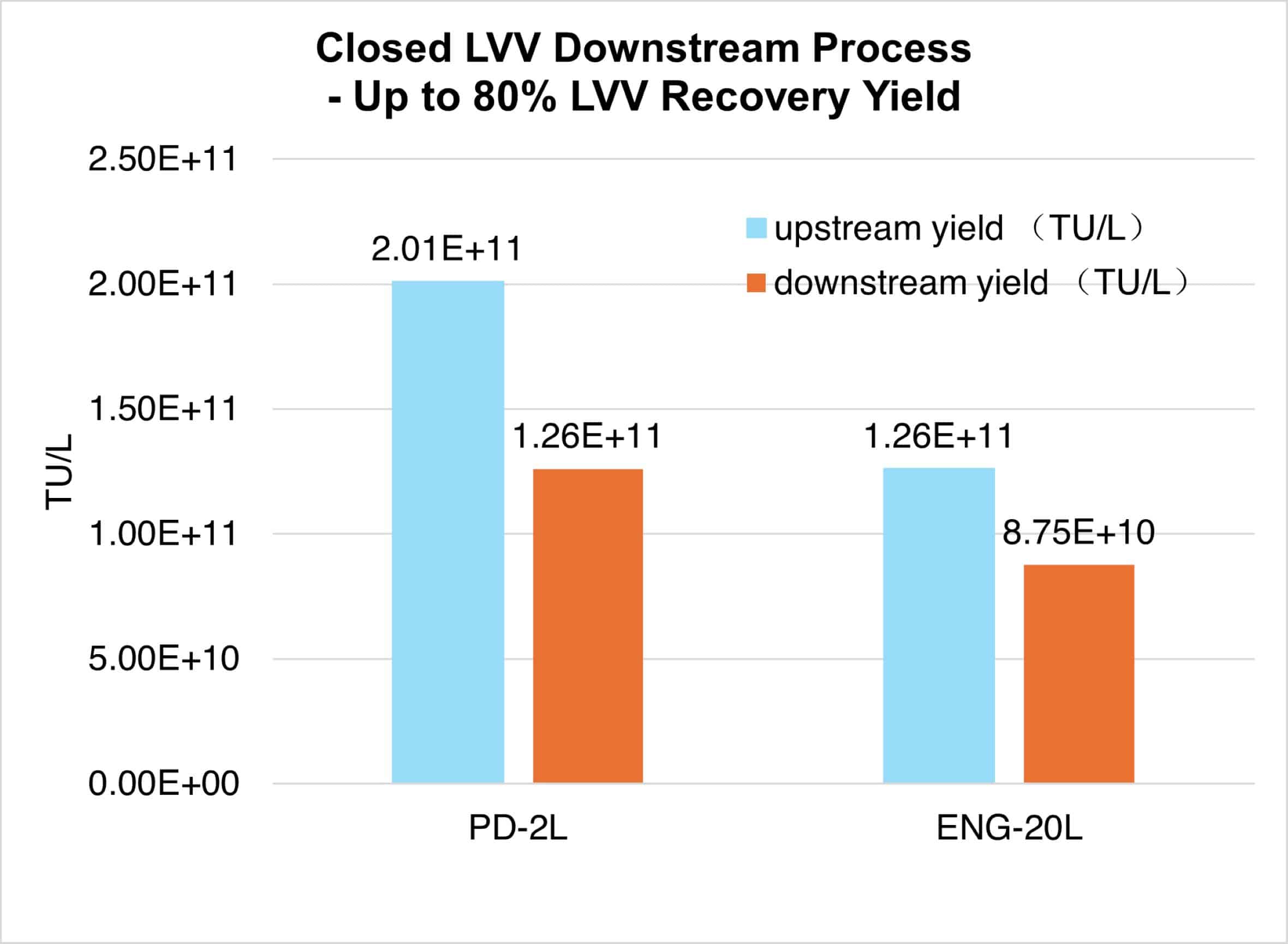
In traditional GMP lentiviral vector manufacturing, the final sterile filtration step often causes significant vector loss due to the large size of LVV particles.
uBriGene’s fully closed downstream purification platform eliminates this step entirely — ensuring sterility while preserving vector integrity. This innovative process achieves up to 80% final recovery yield, maximizing efficiency and reducing production costs.
 Request a quote
Request a quoteFig. 4. CAR-T lentiviral vectors were manufactured using our innovative fully closed downstream system at 2 L and 20 L scales, achieving final recovery rates of 63% and 69%, respectively.
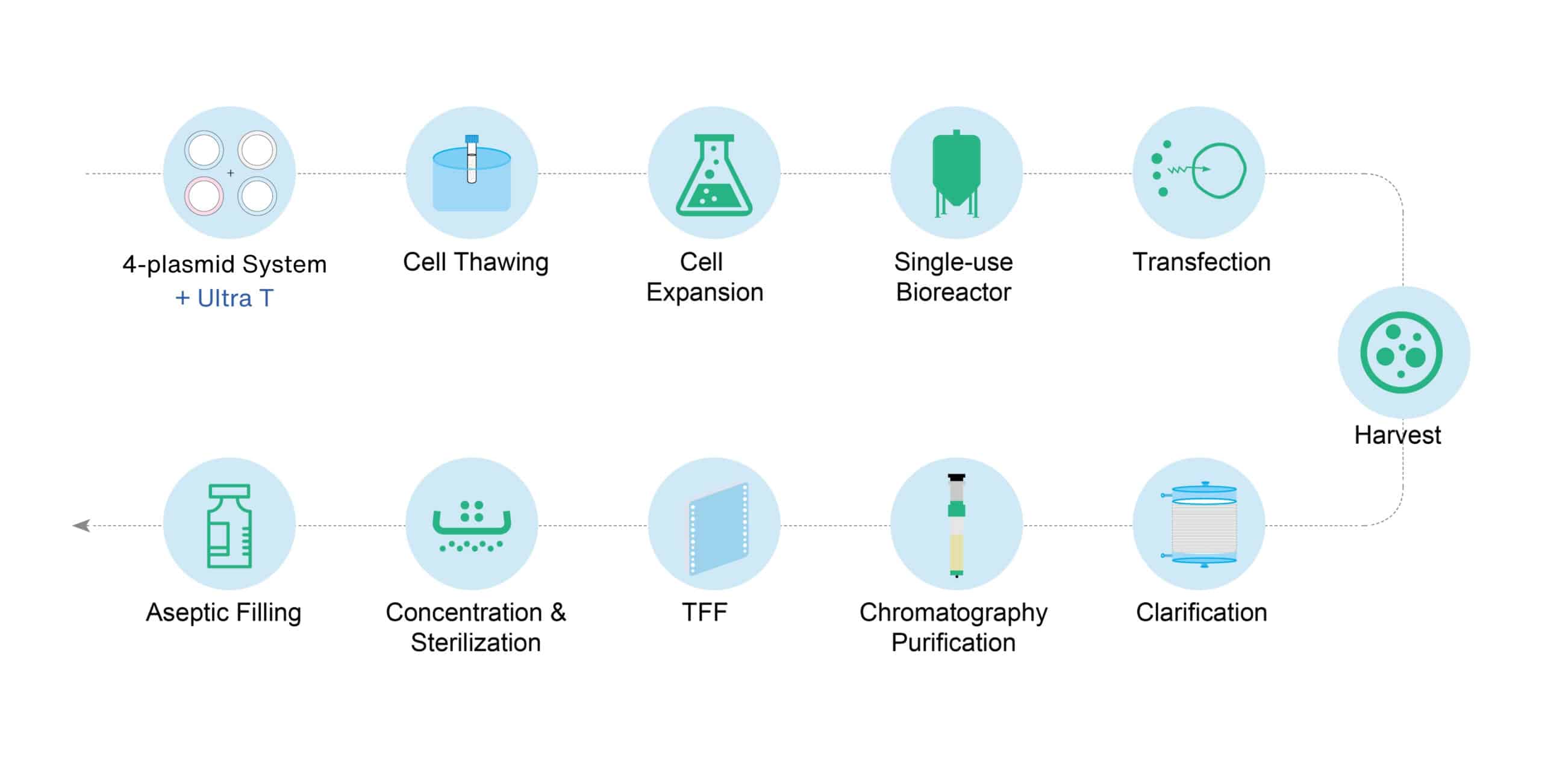
uBriGene brings high-level technical expertise to your lentivirus production plans. Given our experience in developing our own platform, we can rapidly assess your GMP lentivirus production with our current platform to help focus on process and analytical development.
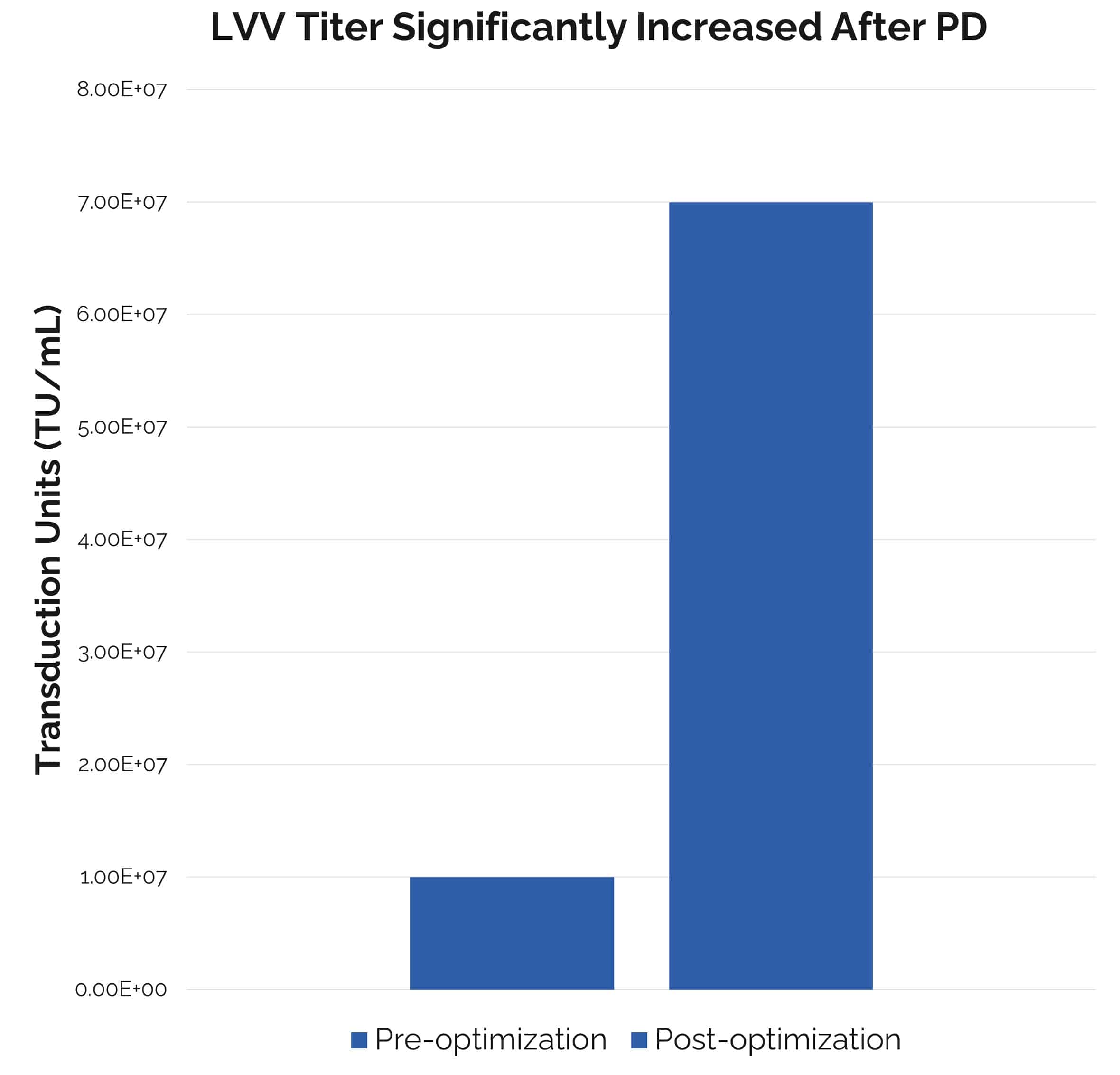
Fig. 2. Lentivirus production platform process development. Through a series of upstream optimizations, the transduction titer has increased by over seven folds.
uBriGene brings deep technical expertise to your lentivirus production programs. Leveraging uBriGene's established LVV production platform, we can accelerate your therapeutic program into the clinic.
Contact Us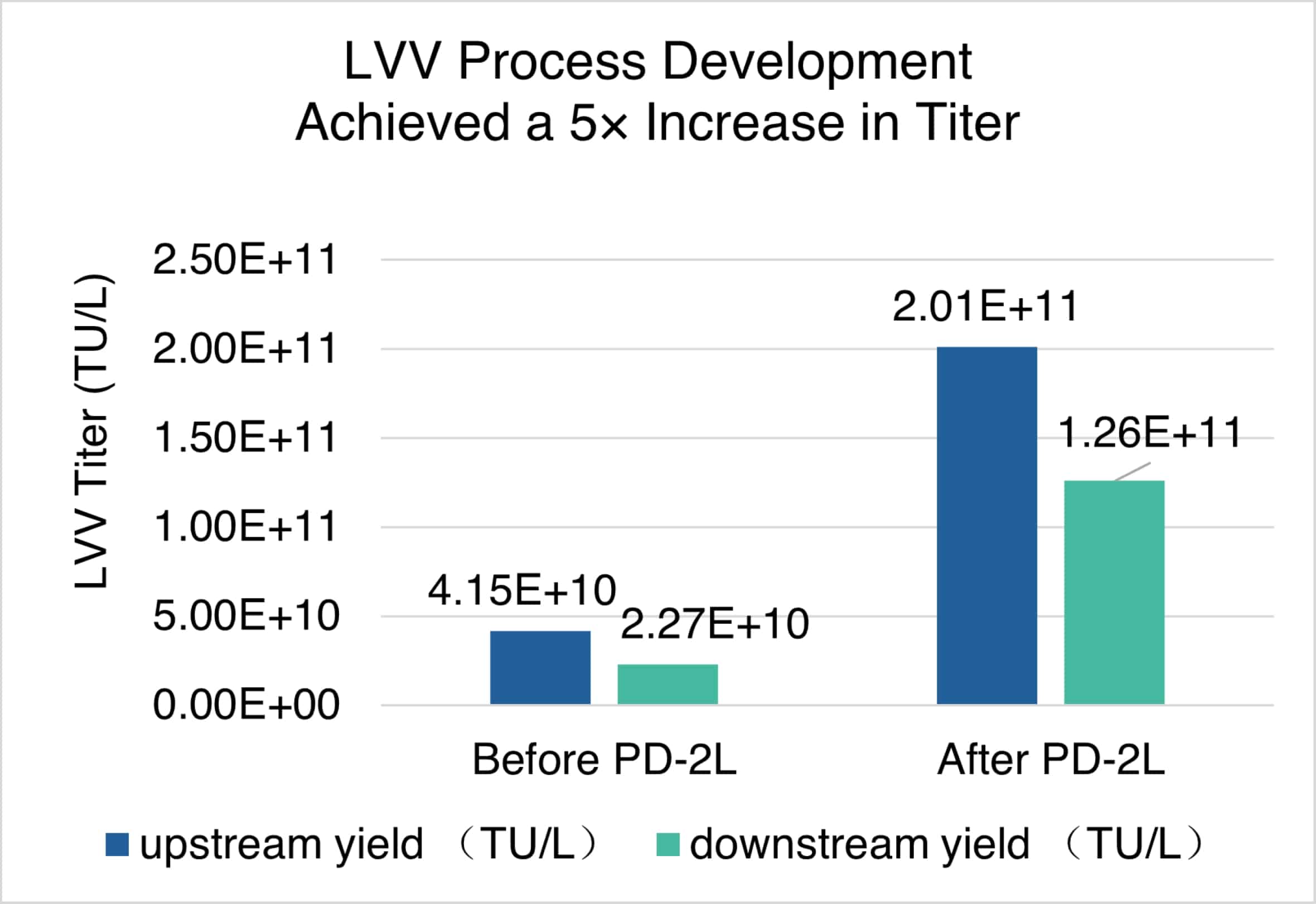
Fig. 5. LVV Turbo™ lentivial vector production platform process development. Through a series of upstream optimizations, the transduction titer has increased by over seven folds.
Discover Lentivirus (LVV Turbo™) production processes and testing
Transforming CAR-T Manufacturing: LVV Turbo™ Breakthroughs Meet Ultra-T Delivery Technology
Learn about CAR-T production process & regulatory guidelines.
Redefining RNA Therapeutics: GMP RNA-LNP Manufacturing You Can Trust
Lentiviral vector remains a leading vector platform for gene-modification of both dividing and non-dividing cell types, and its deployment in the clinical development of cell therapies is an area of active investigation.
We provide the 3rd generation of lentiviral packaging systems for improved safety. If you need 2nd generation lentivirus production, we can also provide that service.
Our GMP-grade lentiviral packaging plasmids are in stock and have an activated DMF with the FDA, which will significantly accelerate IND filing.
As a lentivirus CDMO, leveraging the high-productivity LVV Turbo™ platform, it takes about two months for GMP lentivirus production (excluding time for GMP plasmids production).
We have lentivirus production facilities in both APAC and USA.
Yes, we perform the full spectrum of lentiviral vector quality release testing in-house, except for the RCL assay.
We perform flow cytometry after transducing Jurkat cells with lentivirus using target specific antibody.
We do provide GMP plasmids up to grams in scale.
Yes, uBriGene provides both research grade lentivirus packaging and GMP lentivirus production.
60+ GMP lentiviral vector production projects have been successfully delivered.
The overall yield for a lentivirus production batch is typically around 10%-20%. However, with our closed downstream lentivirus process, our final lentiviral recovery yield can reach up to 80% by eliminating the final sterile filtration.
Low lentiviral vector yields and rising manufacturing costs often result from inefficient upstream processes and purification-related vector loss. uBriGene’s LVV Turbo™ platform combines high-productivity suspension cells with a fully closed downstream process to increase functional titers, enable scalable GMP lentiviral vector manufacturing, and reduce cost of goods for cell and gene therapy programs.
uBriGene’s LVV Turbo™ platform enables scalable GMP lentiviral vector production up to 200L using a fully closed downstream process. It delivers consistent high-yield, high-potency lentivirus from process development through clinical manufacturing.
uBriGene provides end-to-end GMP lentiviral vector manufacturing for cell and gene therapy programs, supporting biotech, pharma, and academic teams from process development through clinical GMP production with scalable, regulatory-ready lentivirus supply.
End-to-end CDMO services with streamlined timelines and cost-effective processes.
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Request Now
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ask the plasmid DNA expert! With over 300 GMP batches in our track record and our cost-effective platform technology, we can help accelerate your clinical programs.