GMP-grade circRNA-LNP manufacturing services ensure high-purity, high-quality RNA drug products, accelerating the progression of therapeutic programs into clinics.

Our platform-based GMP circRNA-LNP manufacturing integrates technical innovations, such as optimized in vitro mRNA transcription, RNA circularization, and RNaseR processing, ensuring over 90% purity and potent therapeutic delivery.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
With our highly productive GMP circRNA manufacturing platform and RNA-LNP formulation technologies, we can fast-track your RNA therapeutic and vaccine programs.
Leveraging our optimized, integrated GMP circRNA-LNP manufacturing platform, we can rapidly implement a plug-and-play approach to deliver potent RNA therapeutic drug products with minimal process development.
The workflow begins with GMP plasmid template production, followed by linearization and purification, then mRNA in vitro transcription and RNA circularization. The purification of circRNA involves the removal of DNA templates and linear mRNA, followed by advanced SEC chromatographic purification and TFF concentration. Finally, the circRNA is encapsulated into LNPs using our proprietary microfluidic technology.
 Request a quote
Request a quoteThe comprehensive circRNA-LNP quality control testing embedded in this platform ensures that each batch meets the highest quality standards, enabling reliable and effective therapeutic solutions that can be safely administered to patients in need.
| Test Item | Method | |
|---|---|---|
| Identity | mRNA sequence identity | NGS/ NanoporeSequencing |
| Content | Concentration | UV absorbance |
| A260/A280 | HPLC/ CE | |
| RNA circularization efficiency | HPLC/ CE | |
| Residual RNase R | Fluorescent probe method | |
| Purity | dsRNA | ELISA |
| Residual DNA template | qPCR | |
| Residual solvents | Residual ethanol(GC-MS) | |
| Potency | Expression of target protein | Cell-based assay |
| Safety | Sterility | Culture method |
| Endotoxin | GEL-CLOT Method | |
| Physical/Chemical Properties | Appearance | Visual method |
| pH | pH | |
*Outsourced testing
| Test Item | Method | |
|---|---|---|
| Identity | Identity of lipids | RP-HPLC |
| Integrity | LNP size and polydispersity | Dynamic light scattering (DLS) |
| Content | Concentration | Fluorescence-based assay |
| Purity | Aggregate quantitation | SEC-HPLC |
| Encapsulation efficiency | Fluorescence-based assay, | |
| Residual solvents | Residual ethanol | |
| Potency | Expression of target protein | Cell-based assay |
| Safety | Sterility | Culture method |
| Endotoxin | GEL-CLOT Method | |
| Physical/Chemical Properties | Appearance | Visual method |
| pH | pH | |
| Osmolality | Osmolality | |
| Subvisible particles | Light blockage methods | |
| Extractable volume | Volumetric method | |
*Outsourced testing

Fig. 1. circRNA template design diagram.
The advantages of membrane matrix in downstream chromatography for plasmid purification include requiring only 30% of the time compared to conventional resin. Therefore, plasmid DNA is manufactured faster and at a much lower cost.
Rhoncus duis elementum suspendisse odio amet arcu tellus. Sapien felis vestibulum euismod eget orci scelerisque id. Non ut sagittis leo et. Et pretium nisl ultrices dapibus sit euismod volutpat elementum.
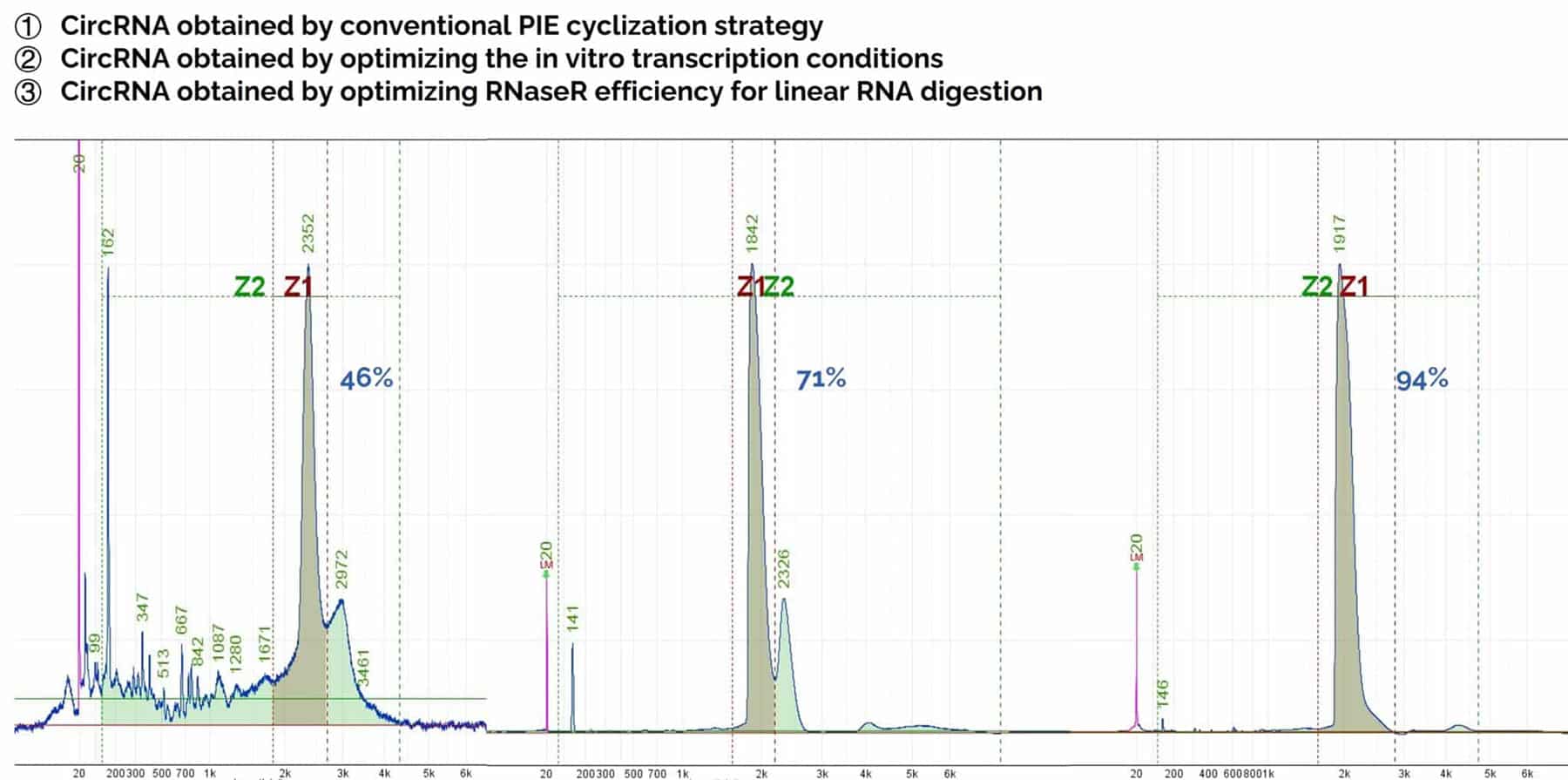
Fig. 2. circRNA purity. Using the conventional PIE strategy, circRNA purity is 46%. After optimizing mRNA in vitro transcription and RNaseR efficiency to remove linear RNA, circRNA purity exceeds 90%.
Rhoncus duis elementum suspendisse odio amet arcu tellus. Sapien felis vestibulum euismod eget orci scelerisque id. Non ut sagittis leo et. Et pretium nisl ultrices dapibus sit euismod volutpat elementum.


Fig. 3. Protein expression of circRNA and mRNA. Same amount of eGFP RNA-LNPs (circRNA-LNP, mRNA-LNP respectively) were added to HEK293 cells. Fluorescent images were taken 24 hours and 48 hours post the treatment. The bottom graph shows the quantification of fluorescence intensity in RNA-LNP treated cells

Lipid nanoparticles containing NeonGreen-teLuc saRNA, circRNA, and linear mRNA (1 pmol / 1E5 cells) were added to HEK293 cells, and Bioluminescence quantification was performed up to 16 days post-treatment.
Redefining RNA Therapeutics: GMP RNA-LNP Manufacturing You Can Trust
Learn about CAR-T production process & regulatory guidelines.
Unlock safer regenerative therapies with Genome-Safe iPSC RPM for clinical success.
Explore RNA drug development fundamentals and the vital role of IVT in manufacturing.
circRNAs exhibit a much longer half-life compared to mRNA, which is beneficial for prolonged protein expression in therapeutic applications.
circRNA translation does not rely on a 5' cap structure, unlike mRNA, which requires this for efficient translation. This enables circRNA to be translated through internal ribosome entry sites (IRES), offering alternative translation mechanisms.
circRNAs can evade immune detection more effectively than mRNA, reducing the likelihood of triggering an innate immune response, which is a common challenge in mRNA-based therapies.
Our circRNA can be delivered in single tubes , suspended in nuclease-free water (1 mg/ml). The circRNA-LNP can be delivered in single tubes , suspended in Tris-buffer or PBS (0.1 mg/ml, RNA concentration).
Typically, circRNA ranges from 100 to 4000 nucleotides in length, but it can be longer if needed.
We can provide research grade and full GMP circRNA for your clinical programs.
Yes, but only a very short scar sequence remains after circularization. However, it has minimal impact on protein expression and immunogenicity.
Extensive expertise with a track record of successfully releasing over 20 GMP batches of mRNA-LNP.
Cost-effective
Reduced timeline
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Ask the Expert
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ask the plasmid DNA expert! With over 300 GMP batches in our track record and our cost-effective platform technology, we can help accelerate your clinical programs.